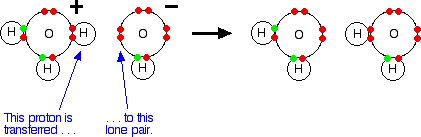
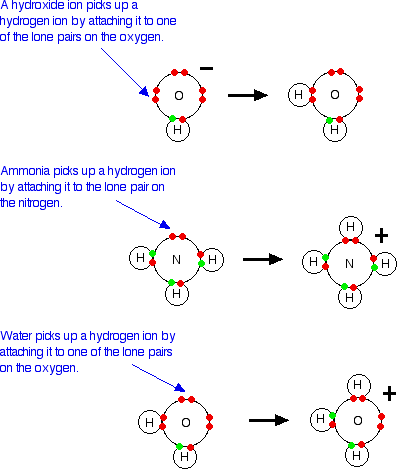
Lewis Acids and Bases - Definition,Properties, Examples, Reactions, Uses, Applications of Lewis acids and Bases.

Acids, Bases and pH. 1. Acids Any compound that GIVES OFF H+ ions in solution Any compound that GIVES OFF H+ ions in solution Ex. HCl H+ and Cl- Ex. HCl. -
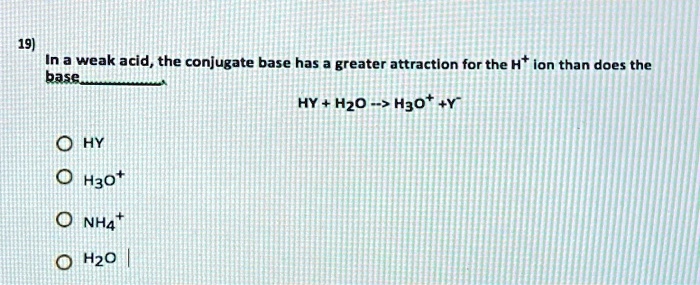
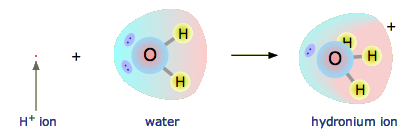
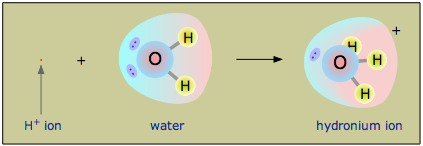
SOLVED: In a weak acid, the conjugate base has greater attraction for the H+ ion than does the base HY + Hzo -> Hgot HY NHA H2O H3o+
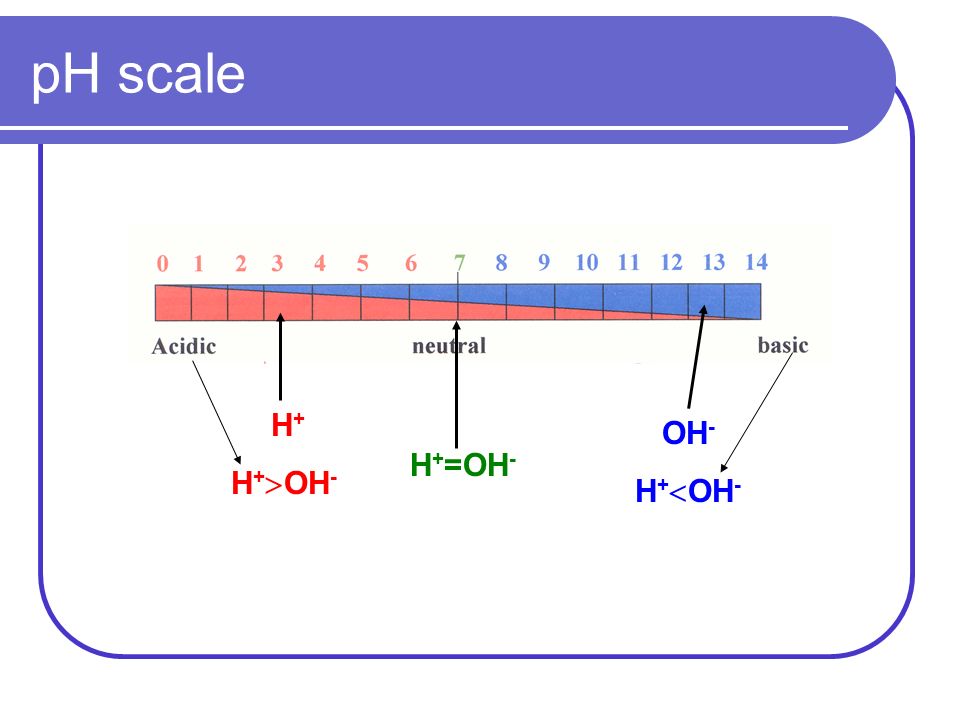
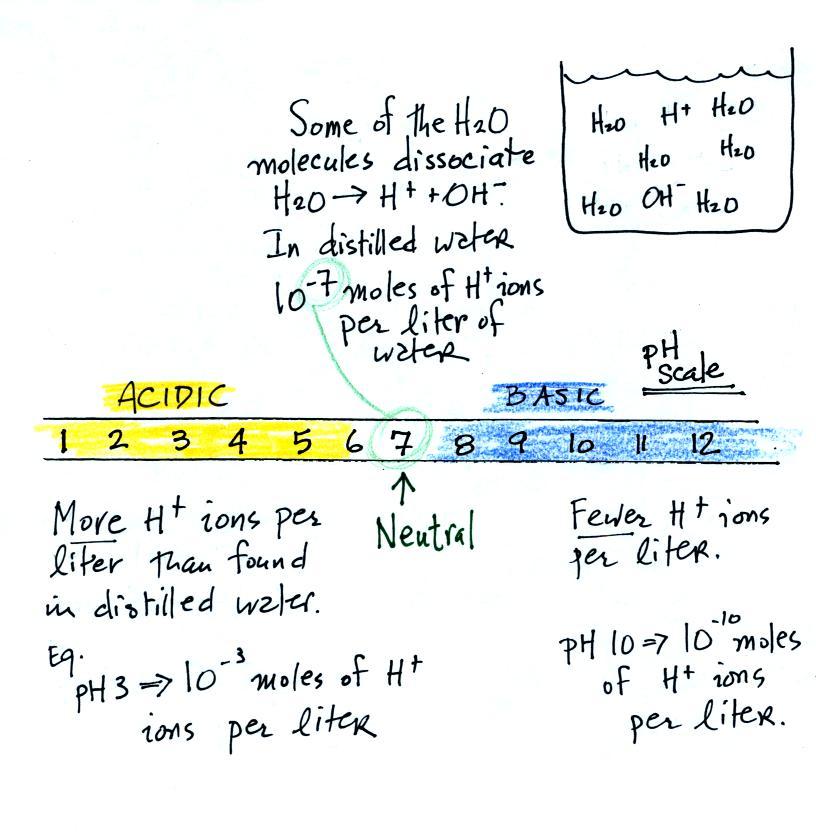
Acid-Base Notes. Acid- Compound that forms hydrogen ions (H + ) when dissolved in water Base – compounds that forms hydroxide ion (OH - ) when dissolved. - ppt download